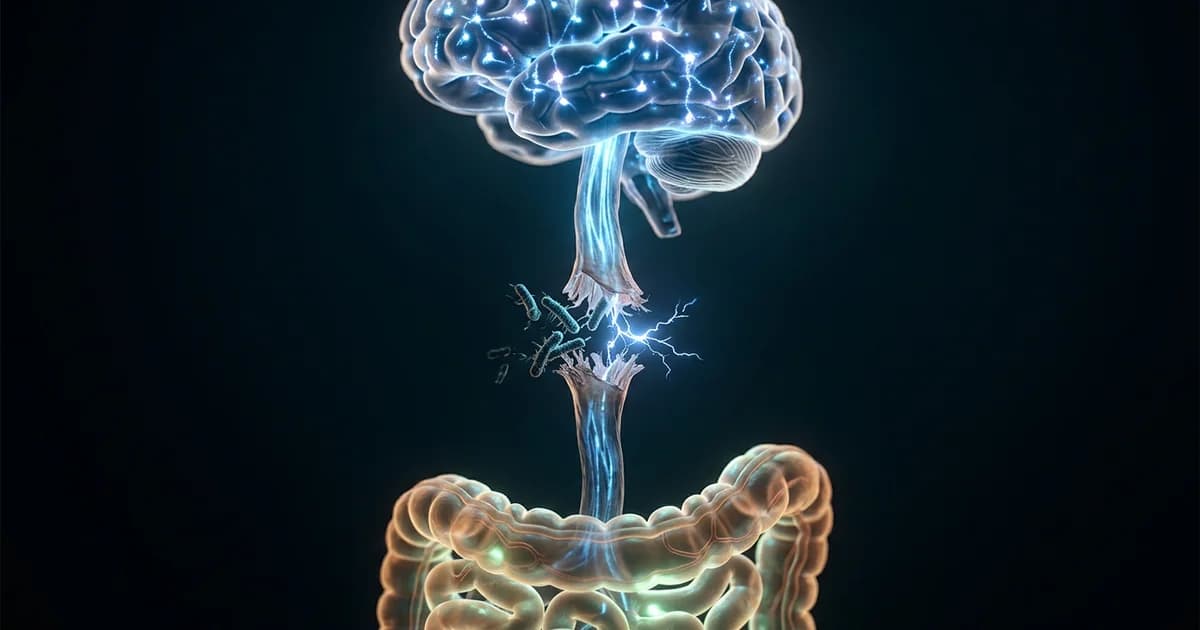
Your gut bacteria are silencing the nerve your memory depends on
Your memory isn't fading because your brain is wearing out. It's fading because something in your gut is quietly cutting the wire between your body and your mind.
A March 2026 study published in Nature by researchers at Stanford Medicine and the Arc Institute identified the exact biological chain reaction: a single bacterial species that accumulates with age, triggers gut inflammation, shuts down your vagus nerve, and blocks the hippocampus (your brain's memory center) from forming new memories. The most striking part? Reactivating that nerve turned forgetful old mice into performers indistinguishable from young ones.
This is not vague "gut health" advice. This is a specific bacterium, a specific nerve, and a specific reversal.
The bacterial saboteur hiding in your gut
The culprit is Parabacteroides goldsteinii, a gut bacterium that quietly increases in abundance as you age. When Stanford researchers led by Christoph Thaiss and Maayan Levy colonized the guts of young mice with this species, something striking happened: the young mice started failing memory tests. They couldn't recognize objects they had seen before. They struggled to escape mazes they had previously navigated easily.
The deficit correlated directly with reduced activity in the hippocampus, the region responsible for forming and storing new memories.
But Parabacteroides goldsteinii doesn't attack the brain directly. It operates through a cascade that scientists had never connected before.
How one bacterium silences your vagus nerve
Here is the three-step mechanism the Stanford team mapped:
Step 1: As Parabacteroides goldsteinii proliferates, it produces elevated levels of medium-chain fatty acids in the gut.
Step 2: Those fatty acids activate a receptor called GPR84 on myeloid immune cells (a type of white blood cell) living in the gut lining, triggering a localized inflammatory response.
Step 3: That inflammation disrupts signaling through the vagus nerve, the longest cranial nerve in your body, which serves as the primary communication highway between your gut and your brain. When vagus nerve activity drops, the hippocampus receives fewer signals, and your ability to form lasting memories degrades.
"The timeline of memory decline is not hardwired," Thaiss explained. "It's actively modulated by the gastrointestinal tract."
This reframes age-related cognitive decline entirely. The problem may not start in your brain at all. It may start in your gut, with bacteria you've never heard of, talking to immune cells through a receptor most doctors don't monitor.
The reversal that stunned researchers
The team then tested whether interrupting this cascade could restore memory function. They stimulated vagus nerve activity in aged mice, and the results were unambiguous: old mice performed at the same level as two-month-old animals on object recognition and maze escape tasks. Not slightly better. Indistinguishable.
Separately, treating old mice with antibiotics for just two weeks, clearing the overgrown Parabacteroides goldsteinii, also restored cognitive abilities.
Two different interventions. Same result: vagus nerve memory function came back online.
Why this matters beyond the lab
Vagus nerve stimulation is already FDA-approved for depression, epilepsy, and post-stroke recovery. The hardware exists. The safety data exists. Extending it to age-related cognitive decline would not require building anything from scratch.
Levy pointed to an even more accessible angle: "Since the gastrointestinal tract is easily accessible orally, modulating gut metabolites is appealing for controlling brain function." Translation: targeting the gut bacteria or their inflammatory byproducts could potentially be done with oral treatments, not surgery.
The critical caveat: these findings are in mice. Parabacteroides goldsteinii is "certainly a member of the human microbiome," but whether it drives the same cascade in humans remains unconfirmed. Clinical trials will need to answer that question.
Still, the mechanism is specific enough to test. And unlike many aging studies that describe correlations, this one mapped a causal chain: bacterium produces fatty acids, fatty acids trigger immune response, immune response silences nerve, nerve silence kills memory. Each link in that chain is a potential intervention point.
What to watch (and what to do now)
If you're already using vagus nerve stimulation devices for stress, you may be unknowingly supporting the same pathway this study identified. Non-invasive vagus nerve stimulators, cold exposure, and even specific breathing techniques (like slow exhalation) activate the same nerve.
The gut-brain axis research is also converging with studies linking mouth bacteria to brain aging and age-reversal therapies targeting cellular mechanisms.
The next time you forget where you put your keys, the answer might not be in your head. It might be 30 feet south, in your gut, where a bacterium you can't feel is slowly turning down the volume on the nerve your brain depends on.
Sources and References
- Nature — Parabacteroides goldsteinii increases with age, produces medium-chain fatty acids that activate GPR84 on gut myeloid cells, triggering inflammation that shuts down vagus nerve signaling to the hippocampus.
- Stanford Medicine — Vagus nerve stimulation in old mice restored cognitive performance to levels indistinguishable from young animals.
- Neuroscience News — The timeline of memory decline is not hardwired; it is actively modulated by the gastrointestinal tract.
- NIH / StatPearls — Vagus nerve stimulation has FDA approval for epilepsy (1997), depression (2005), obesity, post-stroke recovery, and migraine.
Read about our editorial standards →